
Brassica
PGM – (Phosphoglucomutase)
16 % v/v glycerol
Bring 200 grams (= 160 ml) of glycerol (q.c.w. Sigma G-7757) and 800 ml distilled water in a beaker. Mix well and bring to a final volume of 1000 ml with distilled water. Store in the refrigerator.
Sepalyte pH 3-10 (Cat.No. 42008 ProTec Bioseparation)
Ready to hand Sepalyte solutions. Store in refrigerator
Sepalyte pH 3-6 (Cat. No.42006 ProTec Bioseparation)
Ready to hand Sepalyte solutions. Store in refrigerator
Acrylamide/ bis acrylamide 29 : 1 solution
Ready to hand acrylamide solution (q.c.w. Sigma A-3574). Store in refrigerator.
0.1N Sodium hydroxide
4.0 g sodium hydroxide (q.c.w. Sigma S-8045) in 1000 ml distilled water. Add a trace (tip of a small spatula) of sodium azide (q.c.w. Sigma S-8032). Store at room temperature
0,1% w/v riboflavin
0.05 g riboflavin (q.c.w. Sigma R-0508) in 50 ml 0.1 N sodium hydroxide.
Store in the refrigerator for up to 2 weeks.
0.2 M Tris-HCl pH 8.0
Bring 48.4 g Tris (q.c.w. Sigma T-8524) and 0.8 g EDTA (q.c.w. Sigma E-5134) in 1600 ml distilled water. Stir until fully dissolved. Adjust the pH to 8.0 with (± 24 ml) concentrated HCl (q.c.w. BDH 28507). Bring to a final volume of 2000 ml with distilled water. Add a trace (tip of a small spatula) of sodium azide (q.c.w. Sigma S-8032). Store at room temperature.
4 M magnesium chloride
Bring 203-g magnesium chloride hexahydrate (q.c.w. Sigma M-0250) and 150 ml distilled water in a beaker. Stir until fully dissolved and bring to a final volume of 250 ml. Store at room temperature.
Glucose-6-phosphate-dehydrogenase
Dissolve 2000 U in 10 ml water and divide into 0.4 ml in eppendorf vials, equal to 80 U per vial. Store the vials in the freezer.
Gel preparation
9.5 ml 16 % glycerol
2.0 ml Acrylamide/bis 29:1solution
0.7 ml Sepalyte pH 3-6
0.3 ml Sepalyte pH 3-10
0.065 ml 0.1 % w/v riboflavin
0.012 ml TEMED
0.035 ml 10% ammonium persulfate
Add the above reagents and swirl to mix. Pour the gel according to the flap technique and allow polymerizing for at least 4 hours under a light. Store the gels in a sealed bag in the refrigerator for up to 2 weeks.
Sample preparation
Single seeds are placed into a 96 well microplate. To each well, an aliquot * of water is added. The seeds are homogenized using the Terminator for 3 minutes and centrifuged for 10 minutes at 3000 rpm. at 10° C (or at room temperature).
* fraction size 1.25-1.49 mm + 100 ml water.
fraction size 1.50-1.74 mm + 125 ml water.
fraction size 1.75-1.99 mm + 150 ml water.
fraction size 2.00-2.24 mm + 175 ml water.
fraction size 2.25-2.50 mm + 200 ml water.
Electrophoresis
Turn the cooling supply on and set it at a temperature of ± 15°C. Remove the gel from the glass plates. Clean the back of the gel with methanol/ethanol. Place the gel onto the cooling plate with several ml of water. The gel can be divided into four parts. Space the electrodes evenly across the gel, alternating cathode (black electrode) and anode (red electrode), and then place directly onto the gel.
Power settings are for one gel (double the mA and Watts when running two gels).
Prefocusing
Run 1: 600 V–60 mA–12 W–75 V/h
Sample application
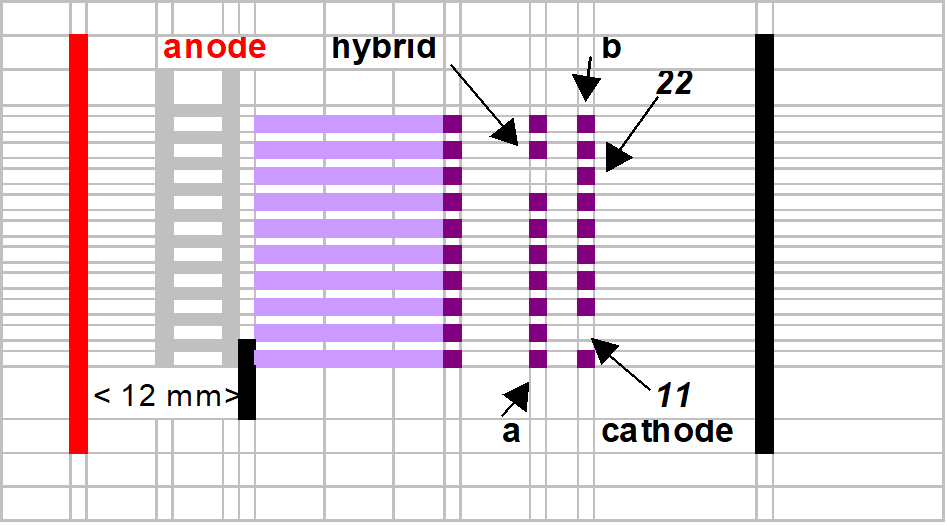
After the prefocusing step, the 96 templates are positioned ± 12 mm from the anode (red electrode). See cartoon image section “gel interpretation. Each sample well is filled with 8 µl of supernatant.
Focusing
Run 2: 200 V–60 mA–12 W–50 V/h
Run 3: 1000 V–60 mA–12 W–700 V/h
After the gel has finished running, remove the gel from the cooling plate and place into an appropriate staining tray.
PGM staining
125 ml 0.2M Tris / HCl pH 8.0
0.2 ml 4 M magnesium chloride
0.50 g α-D-glucose -1-pHospHate disodium salt (Sigma G 7000)
0.05 g NADP (Sigma N-3886)
0.05 g MTT (Sigma M-2128)
0.01 g PMS (Sigma P-9625) (tip of the small spatula)
80 Units Glucose-6-phosphate-dehydrogenase (q.c.w. Sigma G 7750)
Heat up the Tris-HCl solution to ± 37° C. Add the reagents together and mix until fully dissolved and stain the gel at ± 37°C. until the bands can be clearly visualized. Remove the stain and destain the gel in a 2 % glacial acetic acid solution for about 10 minutes and rinse with distilled water. The gel can be air-dried.
Gel interpretation
No international enzyme classification is used.
The neatest band of interest from the anode is genotyped “11”
The farthest band of interest from the anode is genotyped “22”.
The hybrid is genotyped “1/2” in case that the female is 11 and the male 22.
The hybrid is genotyped “2/1” in case that the female is 22 and the male 11
ProTec Bioseparation (Protein Electrophoresis)
